Springfield News Leader: A Local News Leader Providing In-depth Local News And Sports Coverage
The Springfield News-Leader is the most widely circulated newspaper in Springfield, Missouri, and the Ozarks. As of September 2013, the News-Leader has a daily readership of 32,363 and a Sunday circulation of 51,402.
Author:Paula M. GrahamReviewer:Hajra ShannonSep 25, 202135.5K Shares1.1M Views

The Springfield News-Leader is the most widely circulated newspaper in Springfield, Missouri, and the Ozarks. As of September 2013, the News-Leader has a daily readership of 32,363 and a Sunday circulation of 51,402. In the metro region, a single copy costs $2.00, whereas, in the state area, it costs $3.00. On other days of the week, the fee is $2.00. Subscriptions are available in both digital and print formats.
The Springfield Leader started publication in 1867, and in 1933, it combined with the Springfield Daily News to become the Springfield Leader & Press, an afternoon publication. The News & Leader was the morning paper. In 1933, the newspapers relocated to their current location on Boonville Avenue.
In the same year, a new press was built, capable of producing 36,000 sixty-four page sheets each hour. In 1947, the factory was damaged by fire, but with the assistance of local printing companies, a four-page newspaper was out on the streets in a matter of hours.
While the factory was being rebuilt, the newspaper was printed in Tulsa, Oklahoma, for a few months before being trucked to Springfield. Gannett acquired the publication in 1977. In 1987, the morning and afternoon editions were combined to become the Springfield News-Leader.
Marijuana Industry Group Says Missouri Cannabis Companies Filled More Than 4,500 Jobs Since Last Year
600 individuals applied for six new positions at Springfield's newest medical marijuana store, which was authorized to open earlier this year.
According to David Brodsky, director of retail at The Farmer's Wife, this is "a wonderful issue to have." The Springfield-based dispensary celebrated its grand launch the weekend of Aug. 20.
The business also has two additional locations: one in West Plains, which opened on April 20, and another in Mountain Grove, which debuted on May 29. Brodsky estimates that the three shops employ around 25 employees.
Before launching last autumn, John Lopez, an owner of Old Route 66 Wellness, the first dispensary in Missouri's third-largest city, claimed they got more than 500 applications. He claims the locally-owned business currently employs 45 people and is looking for delivery drivers, security guards, and managers.
In November 2020, patients may choose medicinal marijuana at the Old Route 66 Wellness dispensary. Before launching, the dispensary received more than 500 applications, according to John Lopez, the proprietor.
He claimed the business currently employs 45 employees and is hiring delivery drivers, security staff, and management on Sept. 3, 2021. These kinds of figures have been observed throughout Missouri's nascent cannabis sector.
Missouri authorities issued 4,584 state agent ID cards allowing individuals to operate in medical cannabis businesses, according to MoCannTrade, the state's largest organization of marijuana industry participants, which is headquartered in St. Louis.
In early September 2020, when the business was getting ready to serve its first authorized patients, just 200 agent IDs were granted.
In mid-October, the first licensed Missouri dispensaries opened throughout the St. Louis region, and within weeks, shops in southwest Missouri, including Cassville and Springfield, started selling marijuana goods for the first time since voters approved a medical marijuana program in November 2018. "We've certainly tried to be efficient with hiring," The Farmer's Wife's Brodsky said.
"There's a lot of interest in these positions because they're somewhat better-paying occupations, and people want to get in early." Starting dispensary salaries are average, according to Kim Andrews, co-owner and general manager of OzaRX Botanicals in Springfield, which debuted earlier this year.
She said she sometimes hears from Missouri residents who worked in California's cannabis business as candidates want to get in on the "ground floor" of the news industry. She said that they are excited to return to their home state and assist lawful patients.

Running A Marijuana Farming Business | The Highs And Lows Of The Weed Business
As Fda Announces Full Approval For Pfizer Vaccine, CoxHealth Mandates Covid-19 Shots For All Employees
Acting FDA Commissioner Janet Woodcock said in a written statement early Monday that the public may be "extremely confident" that the Pfizer vaccine satisfies the "highest" requirements.
"We realize that for some, the FDA approval of a vaccine may now inspire more confidence to be vaccinated," she added, "although millions of individuals have previously safely received COVID-19 vaccinations."
According to the FDA, the two-dose vaccination may now be sold under the brand name Comirnaty. If you're immunocompromised, the Springfield-Greene County Health Department recommends getting a third COVID-19 vaccination injection.
The Pentagon announced that its 1.4 million active-duty service members would be required to vaccinate within hours of the FDA's announcement, in line with plans announced by the military in early August and expectations among many stakeholders that full FDA approval of at least one COVID-19 vaccine would prompt more government agencies and businesses to require vaccination among their workers.
Missouri's reaction to the approval of the vaccination After the News-Leader contacted their spokesmen about the FDA statement, Gov. Mike Parson's office and the Missouri Department of Health and Senior Services did not immediately reply.
"This clearance confirms what facts and science have proven all along: the vaccine is safe and the greatest resource available to prevent severe disease from COVID-19," Gov. Parson said on Twitter Monday afternoon.
The FDA clearance, according to Gov. Parson, is "excellent news for our state and country," and he hopes it will "relieve any current worries" among Missourians who have not been vaccinated.
Springfield Mayor Ken McClure said in a statement published Monday that the FDA's action "should eliminate any concerns whatsoever regarding the vaccine's safety and efficacy."
"I am hopeful that when people hear this news, they will be encouraged to get vaccinated," McClure said, adding, "for those of you who have been waiting to be vaccinated, now is the time." Cora Scott, the city's director of communications, said Monday that there is no vaccination requirement in place for Springfield municipal workers at this time.
Scott noted that the city manager's office hasn't had time to "consider any modifications for municipal workers as a whole" since the news early Monday morning, but that City Manager Jason Gage's "purview" would allow him to issue a vaccination requirement for city employees without City Council approval.
Meanwhile, the Springfield-Greene County Health Department, according to Scott, may consider requiring vaccines for its own workers "depending on the kind of service they offer to our community."
"The idea behind this step is to get everyone vaccinated," Cox executives said, "and we want to engage with workers as much as possible."
Anyone who has not gotten the first vaccine by the deadline of Oct. 15 will be put on unpaid administrative leave for two weeks while discussions take place on how to resolve any obstacles that prevented them from being vaccinated.
Employees who refuse to get vaccinated and do not have an authorized exemption will eventually have to look for work elsewhere. " Cox said in its statement that approximately 70% of its workers had been vaccinated to date.
More than 90% of doctors are immunized against them. Burrell Behavioral Health officials were "encouraged" by the FDA's approval of the Pfizer vaccine, according to a spokeswoman.
He said that the health system's pharmacies are presently administering the Moderna vaccine, a COVID-19 mRNA vaccine that has not yet received full FDA clearance.
"At this point, we do not intend to require the vaccination for our workers," Burrell officials said in a statement, "but we will continue to evaluate best practices and recommendations from the CDC, public health partners, and colleague health care systems."
Burrell continues to strongly advise all workers, customers, and community people to seek out any vaccination they have access to, as it has done when the vaccine became available last December."

FDA Grants Full Approval To Pfizer Covid Vaccine
FDA Clearance Is "Terrific," According To A Springfield Doctor.
When Dr. Jim Blaine, a renowned local physician who has been involved in the Greene County Medical Society, the DWI Task Force, and other causes for many years, learned of the FDA's statement, he remarked, "I think it's great that that's occurring."
"People who have been reluctant and waited for this time have been our worry." Blaine, who has been vaccinated, said he supports efforts by hospital systems such as CoxHealth to require immunization of local healthcare workers.
"I believe that as health care providers, we owe it to our patients and communities to be vaccinated in order to prevent the pandemic from spreading further," Blaine added. "We, of all people, should be leaders and role models." "Probably the safest vaccination we have... it's definitely also probably the most successful," Blaine said of the Pfizer vaccine.
The Missouri State Medical Association, however, applauded the FDA's decision. In a written statement, the medical group's president, Dr. Alexander Hover, stated, "The Pfizer-BioNTech vaccine has proved to be safe and efficacious."
"It's the simplest approach for Missourians to limit the spread of the Delta variety, decrease COVID hospitalization, and safeguard our communities and families," said the vaccine's manufacturer.
Mercy stated in early July that all of its employees, including those in Springfield, must get vaccinated by Sept. 30 or risk losing their jobs. Since then, at least two anti-vaccine demonstrations near Mercy's Springfield sites have erupted in response to the hospital system's decision.
Katie Towns, director of the Springfield-Greene County Health Agency, said the FDA clearance was "wonderful news" and that the health department is "hopeful" that it will persuade unvaccinated people to receive the vaccine.
At a press conference on Aug. 23, 2021, Springfield-Greene County Director of Health Katie Towns stated that complete FDA approval of the Pfizer vaccine was "excellent news" and that she hoped it would persuade unvaccinated individuals to be vaccinated against COVID-19.
Towns referenced Kaiser Family Foundation research from earlier this year that suggested that with complete FDA clearance, a significant percentage of the unvaccinated population would be more inclined to be vaccinated.
When asked whether the FDA clearance will lead to additional worker vaccination requirements by local companies and other groups, Towns told reporters, "I believe it remains to be seen."
Is The Pfizer Vaccine Still Approved For Emergency Use?
According to the government, the FDA's emergency use permission for the Pfizer vaccine, which was issued in December, would be extended to youngsters aged 12 to 15. Emergency use permission is still in place for 2.7 million immune-compromised people throughout the nation who were recently authorized for a third booster dosage.
Meanwhile, the Biden administration said this week that booster injections would be given starting Sept. 20 for American adults who got the Pfizer or Moderna vaccinations. Vaccinated people should wait eight months after their second dosage before receiving a booster shot, according to government authorities.
The Springfield-Greene County Health Department, according to Towns, will be ready to assist with third booster shots for adults around Sept. 20. The health agency, according to Towns, will wait eight months between the second and booster vaccination shots.
"As demand tells us what the requirements are," Towns told reporters, "we will adapt and be flexible to suit the demands of the community."
What Is The Pfizer Vaccine And How Does It Work?
Unlike previous vaccinations and immunization methods going back to the 1700s, mRNA vaccines like the one produced against COVID-19 do not include any weakened or dead virus or bacterium components, according to Pfizer.
Both the Pfizer and Moderna vaccines contain mRNA or messenger ribonucleic acid. mRNA is responsible for converting DNA information into proteins.
According to MedlinePlus, a service of the US National Library of Medicine, those who get an mRNA vaccination are not exposed to the COVID-19 virus and cannot become infected by the vaccine.
According to the FDA, the mRNA in the Pfizer vaccination is only present in the body for a brief period of time. It is not integrated into a person's genetic material, and it has no effect on that material.
An mRNA vaccine, such as the one approved by the FDA on Monday, is delivered to cells in the human body via lipids, or fatty acids, which are very small particles. The mRNA tells the cells in the body to make the spike protein present on the surface of the new coronavirus that causes infection.
The body's immune reaction starts when human cells produce spike protein, including antibodies that combat the SARS-CoV-2 virus that causes COVID-19 illness.
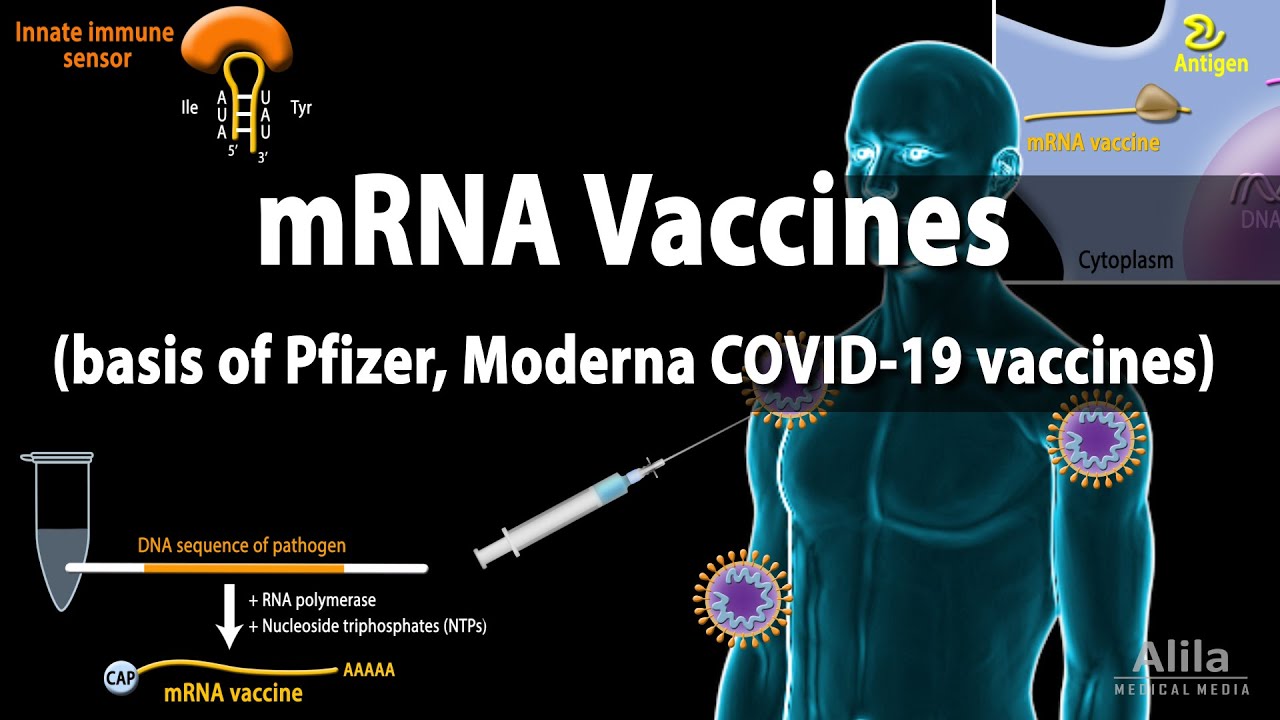
RNA Vaccines (mRNA Vaccine) - Basis of Pfizer and Moderna COVID-19 vaccines, Animation

Paula M. Graham
Author

Hajra Shannon
Reviewer
Latest Articles
Popular Articles
